April 27, 2018
Mayo Clinic is enrolling patients in a phase 1 clinical trial of adipose stem cell treatment for spinal cord injury caused by trauma. The researchers already have approval from the Food and Drug Administration for subsequent phase 2A and 2B randomized control crossover trials.
Lesión de la médula espinal relacionada con un traumatismo
Lesión de la médula espinal relacionada con un traumatismo
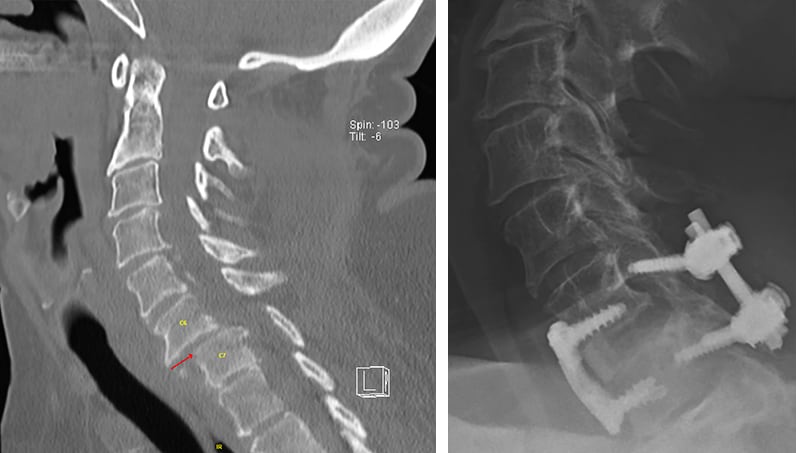
A la izquierda, la tomografía computarizada de un paciente muestra dislocación de la faceta C6-7 y subluxación y fractura del cuerpo vertebral, con la consiguiente lesión de la médula espinal y debilidad en las manos, las extremidades inferiores y los tríceps bilaterales. A la derecha, la radiografía postoperatoria muestra la descompresión anterior y posterior de C6-7 y la fusión instrumentada.
Participants in the phase 1 clinical trial must have experienced a trauma-related spinal cord injury from two weeks to one year prior to enrollment. They will receive intrathecal injections of adipose-derived mesenchymal stem cells. No surgery or implantable medical device is required.
"That is the most encouraging part of this study," says Mohamad Bydon, M.D., a consultant in Neurosurgery specializing in spinal surgery at Mayo Clinic in Rochester, Minnesota, and the study's director. "Intrathecal injection is a well-tolerated and common procedure. Stem cells can be delivered with an implantable device, but that would require surgery for implantation and additional surgeries to maintain the device. If intrathecal treatment is successful, it could impact patients' lives without having them undergo additional surgery or maintain permanently implantable devices for the rest of their lives."
To qualify for the trial, patients must have a spinal cord injury of grade A or B on the American Spinal Injury Association (ASIA) Impairment Scale. After evaluation at Mayo Clinic, eligible patients who enroll will have adipose tissue extracted from their abdomens or thighs. The tissue will be processed in the Human Cellular Therapies Laboratories, which are co-directed by Allan B. Dietz, Ph.D., to isolate and expand stem cells.
Four to six weeks after the tissue extraction, patients will return to Mayo Clinic for intrathecal injection of the stem cells. The trial participants will then be evaluated periodically for 96 weeks.
Mayo Clinic has already demonstrated the safety of intrathecal autologous adipose-derived stem cells for neurodegenerative disease. In a previous phase 1 clinical trial, with results published in the Nov. 22, 2016, issue of Neurology, Mayo Clinic researchers found that therapy was safe for people with amyotrophic lateral sclerosis (ALS). The therapy, developed in the Regenerative Neurobiology Laboratory under the direction of Anthony J. Windebank, M.D., is moving into phase 2 clinical trials.
Dr. Windebank is also involved in the new clinical trial for people with traumatic spinal cord injuries. "We have demonstrated that stem cell therapy is safe in people with ALS. That allows us to study this novel therapy in a different population of patients," he says. "Spinal cord injury is devastating, and it generally affects people in their 20s or 30s. We hope eventually that this novel therapy will reduce inflammation and also promote some regeneration of nerve fibers in the spinal cord to improve function."
Mayo Clinic's extensive experience with stem cell research provides important guidance for the new trial. "We know from prior studies that stem cell treatment can be effective in aiding with regeneration after spinal cord injury, but many questions remain unanswered," Dr. Bydon says. "Timing of treatment, frequency of treatment, mode of delivery, and number and type of stem cells are all open questions. Our hope is that this study can help answer some of these questions."
In addition to experience, Mayo Clinic brings to this clinical trial the strength of its multidisciplinary focus. The principal investigator, Wenchun Qu, M.D., M.S., Ph.D., is a consultant in Physical Medicine and Rehabilitation at Mayo Clinic's Minnesota campus, as is another of the trial's investigators, Ronald K. Reeves, M.D. Dr. Dietz, the study's sponsor, is a transfusion medicine specialist. Also involved is Nicolas N. Madigan, M.B., B.Ch., BAO, Ph.D., a consultant in Neurology at Mayo Clinic's Minnesota campus.
The study team is in discussions with U.S. military medical centers to enroll patients, and discussing additional collaboration with international sites, potentially in Israel or Europe, for future phases of the study.
"At Mayo Clinic, we have a high-volume, patient-centered multidisciplinary practice," Dr. Bydon says. "That allows us to do the most rigorous scientific trial that is in the best interests of our patients."
For more information
Mayo Clinic. Adipose Stem Cells for Traumatic Spinal Cord Injury (CELLTOP). ClinicalTrials.gov.
Staff NP, et al. Safety of intrathecal autologous adipose-derived mesenchymal stromal cells in patients with ALS. Neurology. 2016;87:2230.